Around 80% of disease-causing proteins, including key drivers of many cancers and other serious neurological conditions like Alzheimer’s disease, cannot be targeted by currently available therapeutics. These so-called “undruggable” proteins lack specific surface areas required for treatments such as small molecule inhibitors or antibodies to bind with disease-causing proteins and modulate their function.
However, an alternative therapeutic strategy known as targeted protein degradation has shown the potential to remedy these “undruggable” proteins. Utilizing small molecules known as PROTACs, this strategy harnesses the cell’s waste disposal system to promote the destruction of disease-causing proteins.
What are PROTACs?
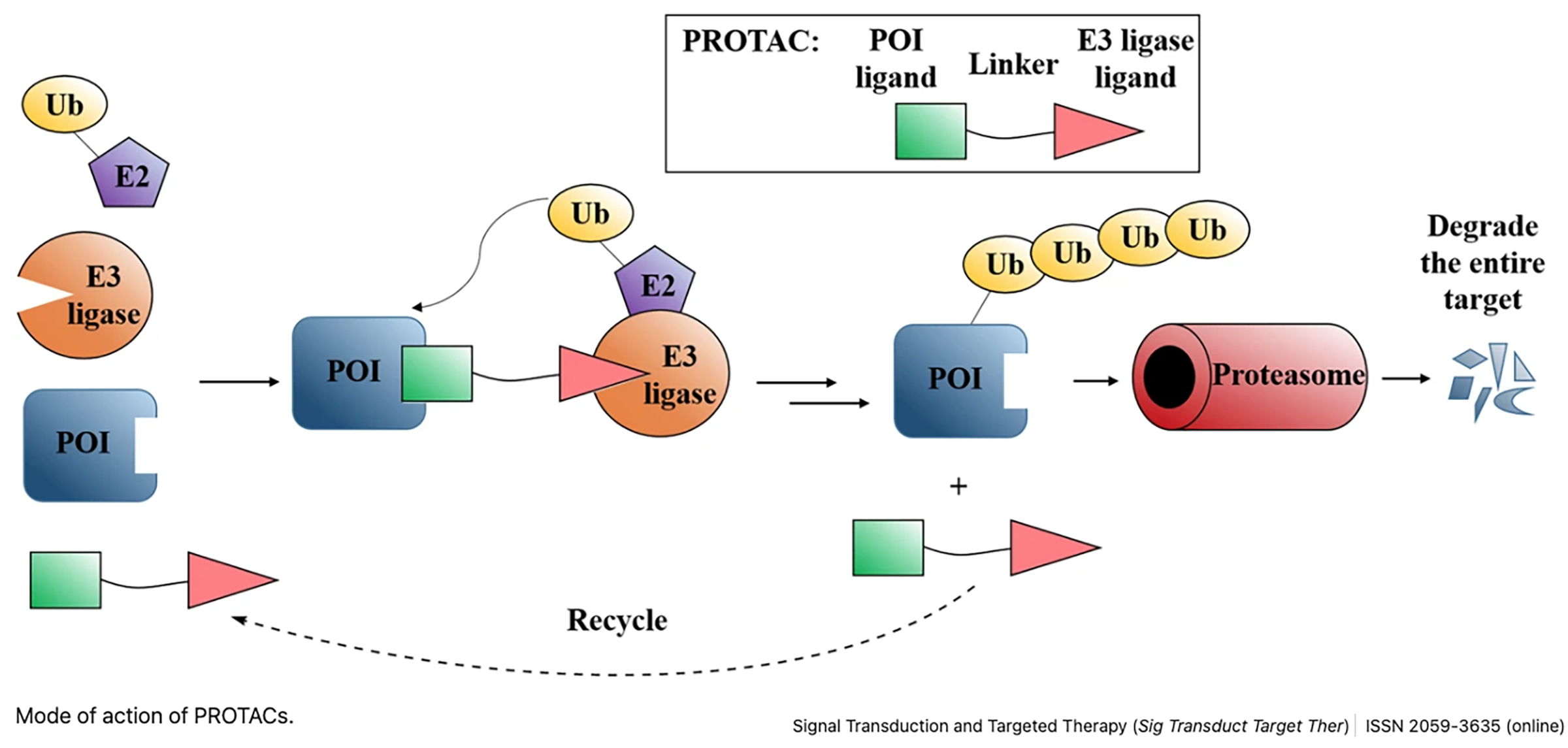
PROteolysis TArgeting Chimeras, or PROTACS for short, are two separate molecules bound together to form a two-headed molecule. One end binds to a ubiquitin ligase, while the other end binds to the “undruggable” protein targeted by pharmacologists. In illustrations, PROTACs are often depicted as dumbbells, but it may be more helpful to think of them as flexible harnesses.
How do PROTACs work?
PROTACs are designed to take advantage of the cell’s waste disposal system that removes unneeded proteins. This system, known as the proteasome, is important for the cell to remove unneeded or damaged proteins and recycle their building blocks to make new proteins. The proteasome plays critical roles in cell growth, management of cellular stress, and the immune system. One end binds to the targeted proteins, while the other end of the molecule binds to the ubiquitin ligase, which then marks the targeted protein for destruction. This lets the cell’s proteasome know that this specific protein can be destroyed. In this way, the body’s regularly occurring mechanisms are co-opted to destroy disease-causing proteins.
What makes PROTACs so unique?
Most therapies are divided between small molecule inhibitors or therapeutic antibodies/biologics. However, “PROTACs are small molecules and as such not restricted to targeting surface proteins, however, in contrast to traditional small molecule inhibitors, PROTACs are fundamentally different,” explained Dr. Fischer, “While inhibitors need to achieve an almost perfect degree of target engagement over an extended period of time to exert their pharmacologic effect, PROTACs follow more of a hit and run strategy.”
“Optimized degrader molecules will have fast rates of degradation and relatively short exposure with therapeutic doses that result in complete elimination of the target protein, which can result in a more durable and deeper effect,” he explained. “More importantly, however, small molecule degraders completely eliminate the disease-causing protein and as such can target the non-catalytic activity of enzymes but also scaffolding proteins, and other non-enzymatic targets.”

When will PROTACs be more widely available?
While researchers have demonstrated the potential of PROTACs in the lab, the first clinical trials are just opening. Still, Dr. Fischer is very optimistic, “The technology has rapidly spread, and we can expect to see many more programs entering clinical development. Due to the pioneering work of a growing academic community spearheading this field, the concepts underlying protein degradation are largely public domain and widely available.”
What is the future of PROTACs research?
“I believe the field of targeted protein degradation is here to stay and will significantly expand our repertoire of therapeutic modalities,” said Dr. Fischer. “I also believe it is still in its infancy and many challenges lie ahead of us to broadly deploy this to the more challenging targets.” PROTACs could potentially prove the impossible is possible by allowing scientists to destroy disease-causing proteins that were previously considered beyond their reach.

-----------------------------------------------------------------
Reposted from: https://www.nyas.org/news-articles/academy-news/what-are-protacs-and-how-do-they-treat-diseases/